Definition:
When you prepare a water lemonade, you mix lemon juice, water and sugar, this is an easy thing to do. But, what happens when you want to separate a component from the mixture? Have you tried to unsweeten the limonade without adding water? It is difficult, right? It would be easier if you had pants and shirts, this is because they are physically different. Separation processes are methods used to divide a mixture into two or more components with different concentration. Depending on the nature of the components in the mixture and the phase(s) they present, there is a process that is appropiate.
Most common separation processes:
For the purpose of this subject, distillation concept will be expanded.
Flash distillation:
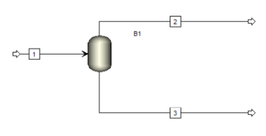
It is one of the simplest separation process. A liquid stream containing several components is partially vaporised in a ”flash drum” at a certain pressure and temperature. This results in two phases: a vapor phase, enriched in the more volatile components, and a liquid phase, enriched in the less volatile components.
The system is called "flash” distillation because the vaporization is extremely fast after the feed enters the drum. Due to the intimate contact between liquid and vapour, the system in the flash chamber is very close to an equilibrium stage.
When you prepare a water lemonade, you mix lemon juice, water and sugar, this is an easy thing to do. But, what happens when you want to separate a component from the mixture? Have you tried to unsweeten the limonade without adding water? It is difficult, right? It would be easier if you had pants and shirts, this is because they are physically different. Separation processes are methods used to divide a mixture into two or more components with different concentration. Depending on the nature of the components in the mixture and the phase(s) they present, there is a process that is appropiate.
Most common separation processes:
- Distillation: It is the separation two immiscible liquids and this because each one has its boiling point.
- Evaporation: It involves the removal of a liquid phase from a solution heat treatment.
- Crystallization: Is the reduction of temperature where the insoluble component crystallizes first.
- Decantation: is the separation of heterogeneous mixtures, consisting of a solid and a liquid or two liquids substances and this separation occurs by density difference.
- Centrifugation: Separating a solid from a liquid through a cintrípeta force is developed during the rotation of the centríga, in short words, accelerates the separation process.
- Precipitation: It is obtaining a solid from a solution.
- Filtration: separate mixtures of an insoluble solid and liquid.
For the purpose of this subject, distillation concept will be expanded.
Flash distillation:
It is one of the simplest separation process. A liquid stream containing several components is partially vaporised in a ”flash drum” at a certain pressure and temperature. This results in two phases: a vapor phase, enriched in the more volatile components, and a liquid phase, enriched in the less volatile components.
The system is called "flash” distillation because the vaporization is extremely fast after the feed enters the drum. Due to the intimate contact between liquid and vapour, the system in the flash chamber is very close to an equilibrium stage.
2015. Ana Botello, Jessica Moctezuma, Lenia Sánchez, Mariana Muñoz, María José Morales
