Antoine´s equation
Antoine´s equation is used to calculate vapour pressure of pure substances at different temperatures.
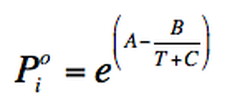
e
(Eq. 3-3)
Where:
A, B and C are established coefficients that can be found in tables, they change depending the substance.
T: Temperature
The units of measurement used depend on the units of the coefficients.
(Eq. 3-3)
Where:
A, B and C are established coefficients that can be found in tables, they change depending the substance.
T: Temperature
The units of measurement used depend on the units of the coefficients.
Raoult´s law
Raoult´s law states that the vapour pressure of each component in an ideal solution depends on the vapour pressure of each individual component and its molar fraction. Therefore, the vapour pressure of the dilution is always less than the pure component´s one.
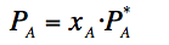
(Eq. 3-4)
Where:
PA=Partial pressure of component A in the mixture/solution.
XA=Mole fraction of component A in the mixture/solution.
P*A=Vapour pressure of the pure component A.
Solved problem using raoult´s law:
Problem:
The water vapour pressure at 20°C is 17.5torr. Consider that the temperature remains constant as glucose (C6H12O6 ) is added to a glass of water, the mole fractions are:
xH2O = 0.8
xC6H12O6 = 0.2
Caculate the final vapour pressure using Raoult's Law.
Solution:
Knowing that the solvent is water and the solute is glucose we have:
P*A=17.5 torr
XA=0.8
Then:
PA=XA·P*A
PA= 0.8·17.5torr = 14torr.
The solution vapour pressure is 14 torr.
The water vapour pressure at 20°C is 17.5torr. Consider that the temperature remains constant as glucose (C6H12O6 ) is added to a glass of water, the mole fractions are:
xH2O = 0.8
xC6H12O6 = 0.2
Caculate the final vapour pressure using Raoult's Law.
Solution:
Knowing that the solvent is water and the solute is glucose we have:
P*A=17.5 torr
XA=0.8
Then:
PA=XA·P*A
PA= 0.8·17.5torr = 14torr.
The solution vapour pressure is 14 torr.
2015. Ana Botello, Jessica Moctezuma, Lenia Sánchez, Mariana Muñoz, María José Morales
