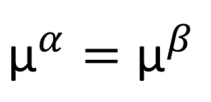Chemical potential
It is related with de Gibbs free energy, it shows us how this energy will vary with the number of moles. (Geochemistry, M.White)
Difference in chemical potential is the driving force for mass transfer.
Difference in chemical potential is the driving force for mass transfer.
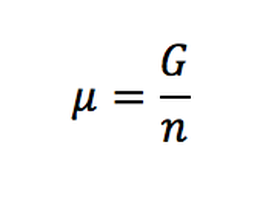
(Eq. 1-9)
Where:
μ= chemical potential
G= Gibbs free energy
n=quantity of substance
This formula is only for one component system.
Where:
μ= chemical potential
G= Gibbs free energy
n=quantity of substance
This formula is only for one component system.
Characteristics of the chemical potential
- Tendency to diffuse: is the tendency of particles to escape or diffuse into the atmosphere.
- Rate of change: this is when carrying out a reaction and a thermodynamic function determines the balance of this.
- Characteristic energy: is the energy change experienced by the system by adding a particle when it is at constant volumen and entropy.
And the thermodynamic criterion of phase equilibrium is:
Fugacity
What is fugacity?

Have you ever been in a room full of people and want to change to another one with less people in it? Well, that is an example of fugacity, you being the representation of any substance (solid, liquid or gas), and fugacity being your will to change from a place full of people to the one that is emptier.
The scientific definition is the tendency of a substance to escape from a phase, a phase being the atmosphere, a lake, the sea, soil, etc. Because it refers to the force made by particles of a substance on the surface between phases, fugacity is measured in pressure units such as atmospheres, Pascals and Bars. Fugacity is also defined as the driving force for mass transfer because
The scientific definition is the tendency of a substance to escape from a phase, a phase being the atmosphere, a lake, the sea, soil, etc. Because it refers to the force made by particles of a substance on the surface between phases, fugacity is measured in pressure units such as atmospheres, Pascals and Bars. Fugacity is also defined as the driving force for mass transfer because
Why is it useful?
It is used to understand the equilibrium between enviromental phases and the movement of substances between them. In order to have equilibrium, the substances in the phases involved need to have the same fugacity, or there will be substance transportation according to the concentration gradient, from a place with higher concentration to a lower concentration until their fugacities are equal.
Equilibrium: f1 =f2 (Eq. 1-11)
Equilibrium: f1 =f2 (Eq. 1-11)
Important characteristics:
-Linearly proportional to concentration
-Used to understand and predict the fate of a substance in the environment, the direction of in which the substance tends to diffuse to reach equilibrium
-Used to understand and predict the fate of a substance in the environment, the direction of in which the substance tends to diffuse to reach equilibrium
fugacity coefficient(φ)
It helps us to see how ideal is the substance we are working with.
If:
φ=1 The substance is ideal
φ <1 There are stronger intermolecular forces, less tendency to scape
φ >1 The substance has more tendency to scape
To calculate this coefficient we have:
If:
φ=1 The substance is ideal
φ <1 There are stronger intermolecular forces, less tendency to scape
φ >1 The substance has more tendency to scape
To calculate this coefficient we have:
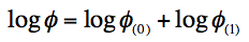
(Eq. 1-12)
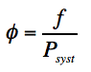
(Eq. 1-13)
2015. Ana Botello, Jessica Moctezuma, Lenia Sánchez, Mariana Muñoz, María José Morales