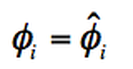Lever rule
If a mixture exists in more than one phase, the Lever rule is used to calculate the amount of each component in each phase. It is known that, according to mass balance, the total amount of substance (F) is the sum of tha vapour phase (V) and the liquid phase (L):
F=V+L (Eq. 3-5)
Then:
1 = V/F + L/F
If we consider a binary mixture composed of componen A and B, the mass balance for component A is:
F=V+L (Eq. 3-5)
Then:
1 = V/F + L/F
If we consider a binary mixture composed of componen A and B, the mass balance for component A is:
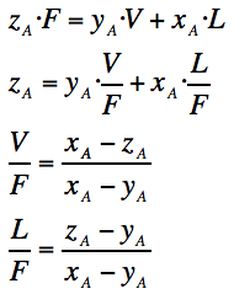
Where:
ZA=Mole fraction of component A in the total mixture
YA=Mole fraction of component A in the vapour phase
XA=Mole fraction of component A in the liquid phase
(Eq. 3-6)
(Eq. 3-7)
ZA=Mole fraction of component A in the total mixture
YA=Mole fraction of component A in the vapour phase
XA=Mole fraction of component A in the liquid phase
(Eq. 3-6)
(Eq. 3-7)
Experimental equilibrium data is presented as function of temperature or pressure and x,y (where x and y are molar fraction of a component in liquid and vapour phase, respectively). In a biinary mixture, the component that is represented in the graph is the most volatile one.
Based on Raoult´s rule and Lewis fugacity rule, the following assumption can be made:
In order to have equilibrium:
In order to have equilibrium:
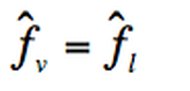
(Eq. 3-8)
According to the fugacity equation for a component in a mixture:
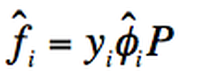
(Eq. 3-9)
Lewis fugacity rule dictates that:
Then, for an ideal gas/ ideal solution :
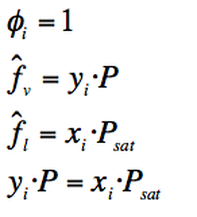
Where:
yi=Mole fraction of component i in the vapour phase
xi=Mole fraction of component i in the liquid phase
P= Low pressure where the behaviour is ideal.
(Eq. 3-10)
yi=Mole fraction of component i in the vapour phase
xi=Mole fraction of component i in the liquid phase
P= Low pressure where the behaviour is ideal.
(Eq. 3-10)
2015. Ana Botello, Jessica Moctezuma, Lenia Sánchez, Mariana Muñoz, María José Morales