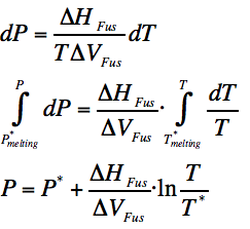State postulate of pure component
"All intensive thermodynamic properties can be determined from two independent intensive properties. " (Onkar, Singh 2009, p. 213)
|
Degrees of Freedom
Number of independent intensive properties. |
Gibbs Phase Rule
F=C-P+2 C->number of components P->number of phases |
Location of the phase boundaries
The slope of the phase boundarie depends on the phase transition occuring, due to the different changes happening on the process. The slope is defined as the change of pressure as temperature is changing (dP/dT) and, because the system is at equilibrium at this points, the following can be deduced:
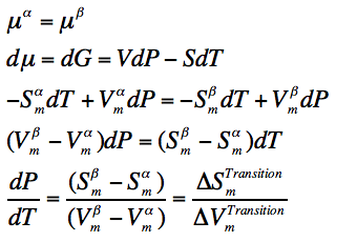
(Eq. 2-1)
As the variation in molar volume depends on the phase transition happening, the steepnes of the slope is different for each transition.
Solid-Liquid boundary
Melting is accompannied by a ΔH of fussion. The molar entropy of melting at an specific temperature T is ΔHfus /T
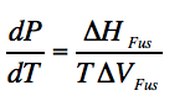
(Eq. 2-2)
As the distance between molecules is a little bit bigger in a liquid than in a solid, so the ΔV is positive but small. As the the ΔH of fussion is also positive, it makes a positive and steep slope.
Assuming that the quotient of variation of enthalpy and volume is so small that is almost constant, the following statement can be done:
Liquid-Vapour boundary
Following the example of the solid-liquid boundary we get:
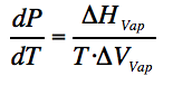
(Eq. 2-4)
A substance in vapour state occupies much more volume than when it is in liquid phase, therefore the ΔV is positive and large. As the the ΔH of vaporization is also positive, it makes a positive slope but less steeper than the solid-liquid boundary.
Assuming an ideal behaviour, we take the state equation (PV=RT) and replace V with RT/P:
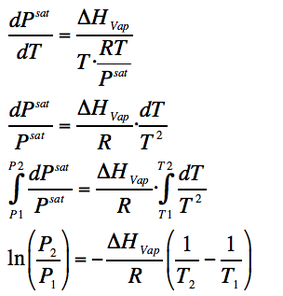
(Eq. 2-5)

Through this process, we get the Calusius-Clapeyron equation (Eq. 2-5).
Solved example using Clausius-Clapeyron equation:
Problem: Calculate the boiling point of water at an altitude of 275m (P@275m=98.7kPa)
Solution:
First, identify the data that the problem indicates and what are you looking for.
Known data:
-P2 =98.7kPa.
-R=0.4619 kPa·m3 / kg·K
-ΔHvap=2257 kJ/kg
Reference state:
-P1 =101.325kPa (1 atm)
-T1=100°C= 373 K (Remember to use absolute temperatures only)
Unknown data:
T2=?
From the equation 2-5, we get:
Solved example using Clausius-Clapeyron equation:
Problem: Calculate the boiling point of water at an altitude of 275m (P@275m=98.7kPa)
Solution:
First, identify the data that the problem indicates and what are you looking for.
Known data:
-P2 =98.7kPa.
-R=0.4619 kPa·m3 / kg·K
-ΔHvap=2257 kJ/kg
Reference state:
-P1 =101.325kPa (1 atm)
-T1=100°C= 373 K (Remember to use absolute temperatures only)
Unknown data:
T2=?
From the equation 2-5, we get:
Solid-Vapour boundary
We do the same with the sublimation enthalpy:
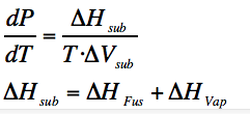
(Eq. 2-6)
(Eq. 2-7)
The slope is also positive. In this case, the ΔH is larger than the other two (as shown by equation 2-7) but also the ΔV is larger as the variation in volume is bigger. This predicts a steeper slope for the sublimation curve than the vaporization one at similar temperature.