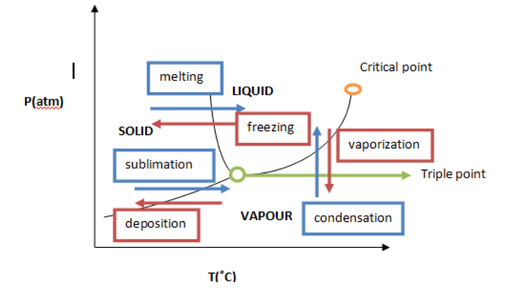
First, some definitions:
To introduce this topic, it is important to define some concepts:
Phase diagram: Map of temperature versus pressure at which each phase of a substance is more stable.
Phase: for of matter that is uniform throughout in chemical composition and physical state.
Phase transition: Spontaneous conversion of one phase into another, and occurs at a specific temperature for a given pressure.
Homogeneous: one phase.
Heterogeneous: two or more phases.
Phase diagram: Map of temperature versus pressure at which each phase of a substance is more stable.
Phase: for of matter that is uniform throughout in chemical composition and physical state.
Phase transition: Spontaneous conversion of one phase into another, and occurs at a specific temperature for a given pressure.
Homogeneous: one phase.
Heterogeneous: two or more phases.
The diagram above is a phase diagram. In it is shown the phases of a substance at different pressure and temperature. The barriers between each phase represent phase transition, the change of phase, and are called phase boundaries. If the conditions at which the substance is on a boundarie, at that moment two or more phases coexist in equilibrium.