|
Did you know that a system can be in steady state without being in equilibrium?
|
What is steady state?
Steady state is reached when the variables don´t change throughout time. Nevertheless, there can still be a driving force present, as it can be concentration gradient, temperature, pressure, etc.
|
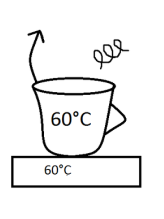
In the picture above is an example of steady state because the temperatures are the same, but it has the tendency to change because the atmospheric temperature is different than the system´s temperature.
|
|
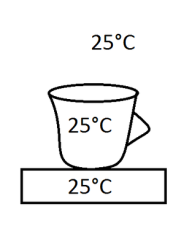
In this picture is an example of equilibrium because all temperatures are the same and the system don´t have the tendency to change.
|
|
Types of equilibrium
Equilibrium can be reached in 4 different ways
- Thermal equilibrium: When the systems temperature and the temperature of the surroundings is the same
- Mechanical equilibrium: When there are no unbalanced forces within the system and between the system and the surrounding. The system is also in mechanical equilibrium when the pressure throughout the system and between the system and surrounding is the same[1].
- Chemical equilibrium: When there are no chemical reactions going on within the system or there is no transfer of matter from one part of the system to another due to diffusion.
- Thermodynamical equilibrium: When the system is in thermal, mechanical an chemical equilibrium at the same time.