Concepts before we start:
Intensive properties: Properties that DON´T depend on the amount of matter (k)
Extensive properties:Properties that DO depend on the amount of matter (K)
Before getting involved in mixtures concerning chemistry, it is important to consider non-reactiv mixtures, binary mixtures.
In a mixture different types of intermolecular interactions are involved, the species that are involved lose their nature, contributing to the properties of the mixture. Also, the characteristics of the product depend on the amount of each component it contains.
Partial molar quantities: Contribution of one component to the property of the total mixture.
-E.g. Partial pressure
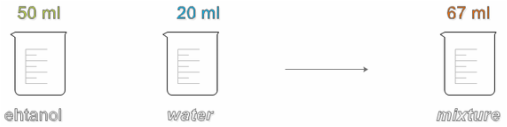
Partial molar volume:
You should be wondering what happened with the 3 ml missing in the flask with the mixture.
The mass is maintained but the volume changes, this because of the intermolecular interactions between the molecules of water and the ones of ethanol.
Maxwell Relation: Mathematically describe any intensive themodynamic property in terms of partial derivations of two independent intensive properties.
GIBBS -DÜHEM THEOREM FOR EQUILIBRIUM SYSTEMS:
Equation that gives useful relationships between the partial molar properties of different species in a mixture.
criteria for chemical equilibrium in multicomponent systems
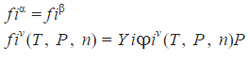
(Eq. 3-1)
Methods to calculate fugacity in mixtures:
The main methods are:
Lewis fugacity rule:
The main methods are:
- Lee Kesler
- EOS (Van der Waals)
Lewis fugacity rule:
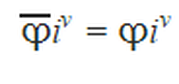
(Eq. 3-2)
2015. Ana Botello, Jessica Moctezuma, Lenia Sánchez, Mariana Muñoz, María José Morales
