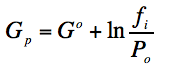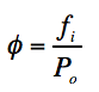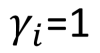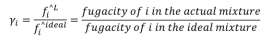Fugacity calculations for gases:
In the section Enthalpy, Entropy and Gibbs Free Energy, equations for the Gibbs free energy behaviour during phase transition was specified for gases (see equations 1-6, 1-7 and 1-8). It was concluded that for 1 mol of an ideal gas, the Gibbs Free Energy is calculated as:
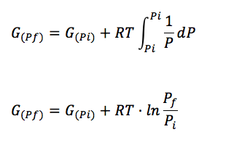
(Eq. 1-8)
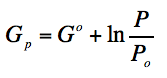
If Pi=P° and Pf =P
Then we have the molar gibbs energy of an ideal gas:
(Eq. 1-14)
For a real gas we replace in the last equation P by the effective pressure called “Fugacity”
Then we have the molar gibbs energy of an ideal gas:
(Eq. 1-14)
For a real gas we replace in the last equation P by the effective pressure called “Fugacity”
We have the fugacity coefficient (Φ) that is the adjusted pressure due to lack of ideality
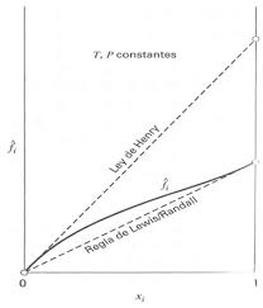
Fugacity calculations for liquids:
What is activity coefficient?
It´s the correction factor to calculate the fugacity of components in liquid phase. It depends on the intermolecular interactions present, the amount of substance and the nature of it.
ACTIVITY COEFFICIENT MODELS
Definition:
Activity coefficient models are models that simulates the behavior of chemical components in a mixture according to their nature, considering the fugacity and activity coefficient. In this way, we will see how its behaviour deviates from ideality.
Activity coefficient models are models that simulates the behavior of chemical components in a mixture according to their nature, considering the fugacity and activity coefficient. In this way, we will see how its behaviour deviates from ideality.
how to choose which one to use?
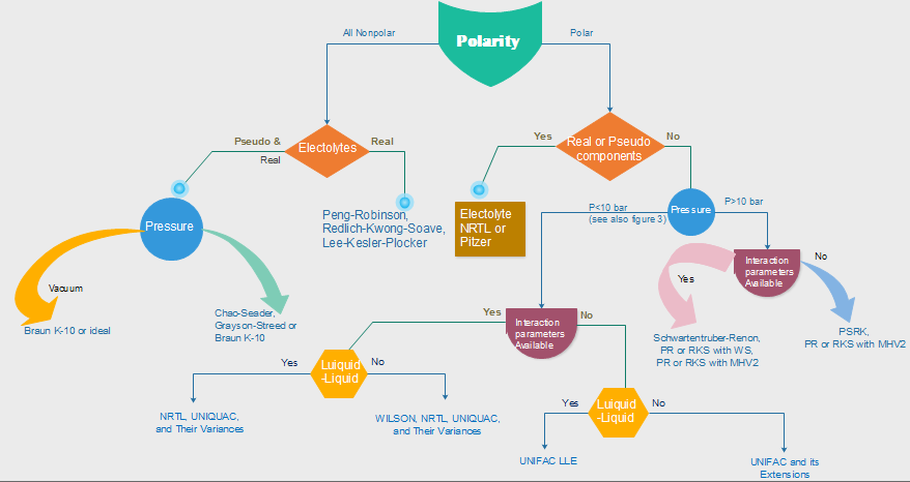
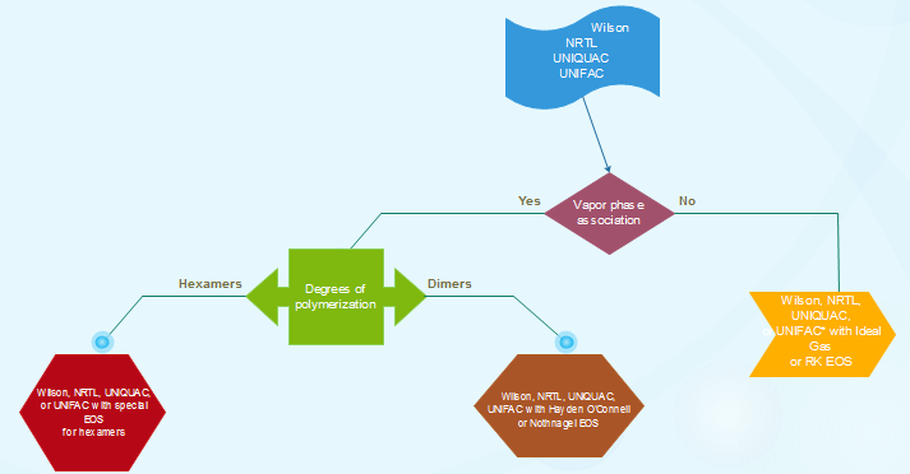
Different activity coefficient models are used for different types of substances, some are best fitted for specific mixtures due to the molecular interactions. Depending on what you have in the mixture, a model is recommended. For your understanding, we have made a flowchart and a comparative table for you to know which models are best for which mixtures.
Figure 1.1. The first steps for selecting physical property methods.
Figure 1.2. Options for vapor-phase claculations with activity-coefficient models.
*Flowcharts made based on the paper "Don’t Gamble With Physical Properties For Simulations" (Carlson, 1996) [6]
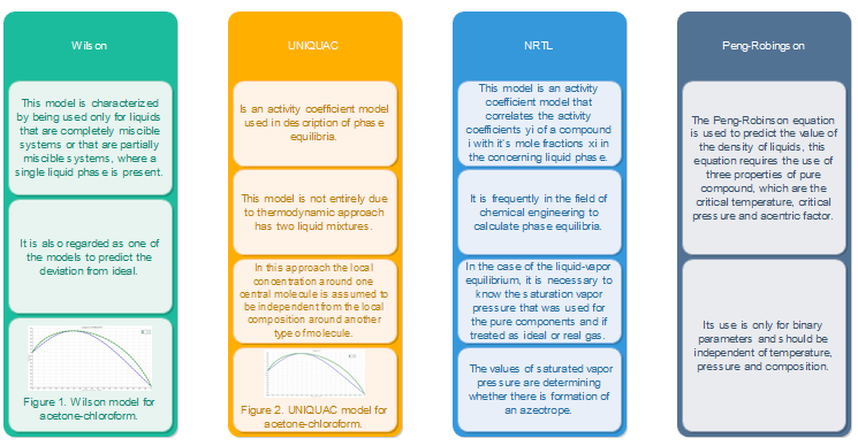
The four main activity coefficient models are: Wilson, NRTL, UNIQUAC and Peng-Robinson. In Figure 1.3 you can find out what are their aims and when to use them.
table 1.3. some activity coefficient models.
2015. Ana Botello, Jessica Moctezuma, Lenia Sánchez, Mariana Muñoz, María José Morales