Enthalpy
The enthalpy change is the heat produced or absorbed during a chemical or physical transformation taking place at constant pressure. (Encyclopedia of Astrobiology, Volume 1)
H=U + PV (Eq. 1-1)
where:
U = internal energy
P=pressure
V=volume
H=U + PV (Eq. 1-1)
where:
U = internal energy
P=pressure
V=volume
Entropy (s)
Have you ever wonder why your earphones always get tangled when you put them inside your pocket? Well, that´s because everything tends to chaos. In natural processes, the force that causes this is called ENTROPY. This very important concept is the measure of disorder, the driving force that causes nature to tend to maximum disorder, due to the stability it gives. A highly organized state has a very low probability of existing while a chaotic one is much more probable.
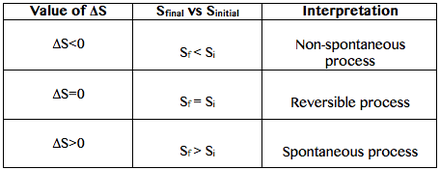
Absolute values of S can be measured, however it´s the variation of entropy the one with real importance:
ΔS=ΔSfinal -ΔSinitial (Eq. 1-2)
How to interprete ΔS?
Absolute values of S can be measured, however it´s the variation of entropy the one with real importance:
ΔS=ΔSfinal -ΔSinitial (Eq. 1-2)
How to interprete ΔS?
When the sum of the ΔS of the surroundings and the one of the system is equal to 0 (zero), the system is at equilibrium.
ΔStotal=ΔSsurroundings + ΔSsystem =0
ΔStotal=ΔSsurroundings + ΔSsystem =0
Simple dayli life Examples
An example of this is when you put lemon juice into water at room temperature. The moment it they interact, both substances mix, without changing the temperature, they just spontaneously mix together. But why doesn´t it naturally unmix? Due to the natural tendency of the process to go to a state of maximum disorder. This process is spontaneous because it became more disordered.
However, when you want to condensate water vapor, you have to apply heat to the system to increase the intermolecular interaction and reduce the distance between particles, organizing them. Here the process became more organized, meaning a negative ΔS, and, because you had to apply work to achieve order, it was non-spontaneous.
However, when you want to condensate water vapor, you have to apply heat to the system to increase the intermolecular interaction and reduce the distance between particles, organizing them. Here the process became more organized, meaning a negative ΔS, and, because you had to apply work to achieve order, it was non-spontaneous.
GIBBS FREE ENERGY
Gibbs free energy is defined as he useful energy, this means the energy that can be used to produce work. To calculate this, we take into account the useful work (enthalpy) and the energy dissipated (entropy), using the following equation:
G=H-TS (Eq. 1-3)
where:
H = enthalpy
T=Temperature (measured in Kelvin)
S=Entropy
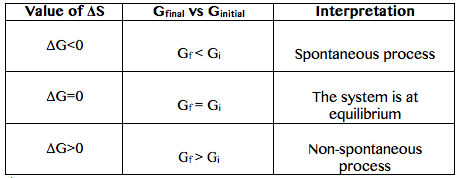
How to interprete ΔG?
The variation of G before and after a process tells us if a it was spontaneous or not.
The ΔG general equation is:
ΔG=ΔH-ΔTS
ΔG=ΔH-TΔS-SΔT (Eq. 1-4)
So, when the conditions are at constant temperature and pressure it can be calculated using the following equation:
ΔG=ΔH-TΔS
G=H-TS (Eq. 1-3)
where:
H = enthalpy
T=Temperature (measured in Kelvin)
S=Entropy
How to interprete ΔG?
The variation of G before and after a process tells us if a it was spontaneous or not.
The ΔG general equation is:
ΔG=ΔH-ΔTS
ΔG=ΔH-TΔS-SΔT (Eq. 1-4)
So, when the conditions are at constant temperature and pressure it can be calculated using the following equation:
ΔG=ΔH-TΔS
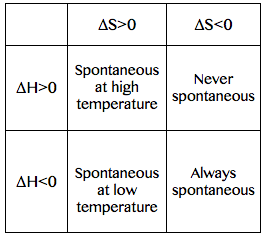
Spontaneity of a process depending on the ΔH and ΔS:
How g varies with p or t?
Mathematical deduction of the Fundamental equation of chemical Thermodynamics:
Putting together:
dG=dH-SdT-TdS
dH=dU+PdV+VdP
dU=TdS-PdV
We get:
dG=dH-SdT-TdS
dG=dU+PdV+VdP-SdT-TdS
dG=TdS-PdV+PdV+VdP-SdT-TdS
dG=VdP-SdT (Eq. 1-5)
Putting together:
dG=dH-SdT-TdS
dH=dU+PdV+VdP
dU=TdS-PdV
We get:
dG=dH-SdT-TdS
dG=dU+PdV+VdP-SdT-TdS
dG=TdS-PdV+PdV+VdP-SdT-TdS
dG=VdP-SdT (Eq. 1-5)
Behaviour of gibbs free energy during phase transition _
During phase transition, the temperature remains constant, dT=0. Substituing in the Fundamental Equation, we get:
dG=VdP
GPf-GPi=VdP
GPf=GPi+VdP
Where:
GPf=Free energy at final pressure
GPi=Free energy at initial pressure
Integrating:
dG=VdP
GPf-GPi=VdP
GPf=GPi+VdP
Where:
GPf=Free energy at final pressure
GPi=Free energy at initial pressure
Integrating:
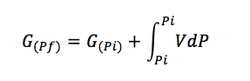
(Eq. 1-6)
To continue the analysis of this equation, it´s important to recognize that for liquids and solids the volume is constant, as for gases it varies according to the equation of state of ideal gases (Eq. 1-7).
a. For liquids and solids (V=constant)
dG=V(Pf-Pi)
b. For gases
a. For liquids and solids (V=constant)
dG=V(Pf-Pi)
b. For gases
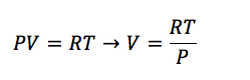
(Eq. 1-7)
Substituing in Eq. 1.2.6:
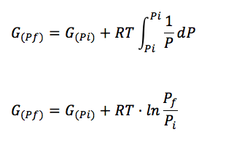
(Eq. 1-8)
2015. Ana Botello, Jessica Moctezuma, Lenia Sánchez, Mariana Muñoz, María José Morales